Legacy Oncology Pipeline
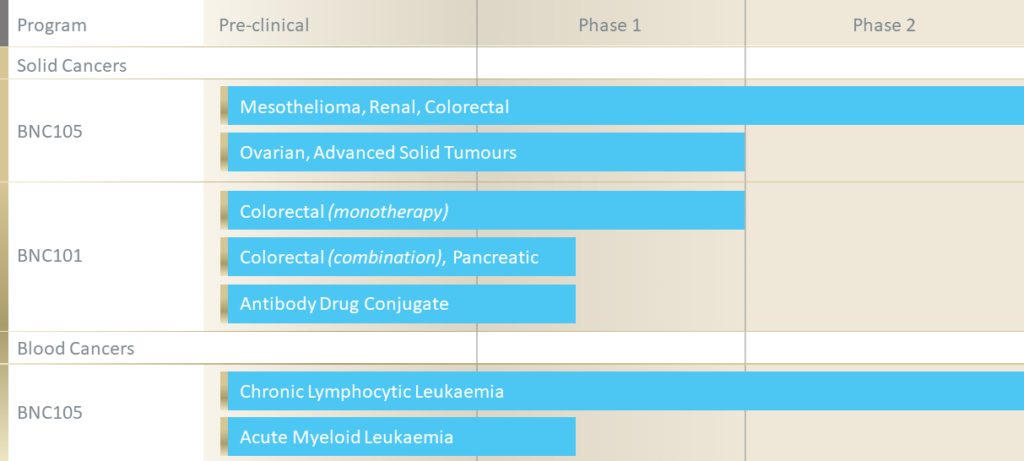
BNC101
BNC101 is a first-in-class humanized monoclonal antibody that targets cancer stem cells (CSC). CSC are a specific group of cells within a tumour thought to play a critical role in tumour initiation, recurrence, and metastasis. BNC101 binds to a receptor that is overexpressed on CSC in solid tumours including colorectal, breast, pancreatic, ovarian, lung, liver and gastric cancers.
CSC are also implicated in resistance to current standard of care treatments. Eliminating CSC, in conjunction with other cancer therapies that address tumour bulk, represents a new concept in cancer treatment that could offer a distinct advantage over existing strategies and a solution for chemo-resistance.
BNC105
BNC105 is a novel small molecule drug being developed as a tubulin polymerisation inhibitor for the treatment of cancer which has multiple modes of anti-cancer action:
- Disruption of tumour blood vessels without affecting blood vessels of normal organs hence starving the tumour of oxygen resulting in the death of thousands of tumour cells
- Directly inducing cancer cell death and inhibition of cancer cell division
- Modulation of the tumour microenvironment when combined with immunotherapy, re-awakening the patient’s immune system to fight the cancer
BNC105 has demonstrated several advantages over classical chemotherapy, allowing it to potentially be used to treat a wide variety of both solid and blood cancers. In clinical studies, BNC105 has been shown to have a good safety profile, shows no additional toxicity when combined with other chemotherapeutics and has been shown to be selectively retained within tumours, improving the selectivity of its action against the tumour.
In pre-clinical studies, BNC105 has shown to be effective when combined with radiation therapy and with other cytotoxic agents generating a greater anti-tumour response. BNC105 is potent in killing leukemic cells and leukemic progenitor cells from patients.
Related Media
PARTNERING OPPORTUNITIES
Bionomics has a lengthy history of successful alliances with industry and academia. We are open to expressions of interest with respect to partnership at all stages of development.